What Is A Vaporizer?
Vaporizers, or vapes as they are commonly known, are electronic devices engineered to release the active ingredients and therapeutic compounds of your desired plant material or extracts. Vaporizers heat your dry herb or concentrates just below the point of combustion to release only the flavor and effects of your material with little to no smoke being produced.
Since no plant matter or concentrate is being burned, no potential carcinogens are being consumed by the user. A 2007 study by the University of California, San Francisco, published in the Journal of the American Academy of Neurology, found that "there was virtually no exposure to harmful combustion products using the vaporizing device" and A 2006 study performed by researchers at Leiden University found that vaporizers were "safe and effective cannabinoid delivery system(s)". The study stated that the amount of THC delivered by vaporizers were equivalent to the amount delivered by smoking. Vapor is much more flavorful and smoother to inhale than smoke, which makes the entire experience more enjoyable and comfortable for the user.
The two categories of vaporizers are desktop and portable. Desktop units are powerful stationary devices made for at-home users, while portable vapes are engineered for on-the-go consumption. As vaporizer technology advances, certain portable units can now achieve the vapor quality of desktop vaporizers. We’ll further unpack the differences between portable and desktop vapes later on. For now, let’s take a look at the history of vaporizers to see just how we got to this point.
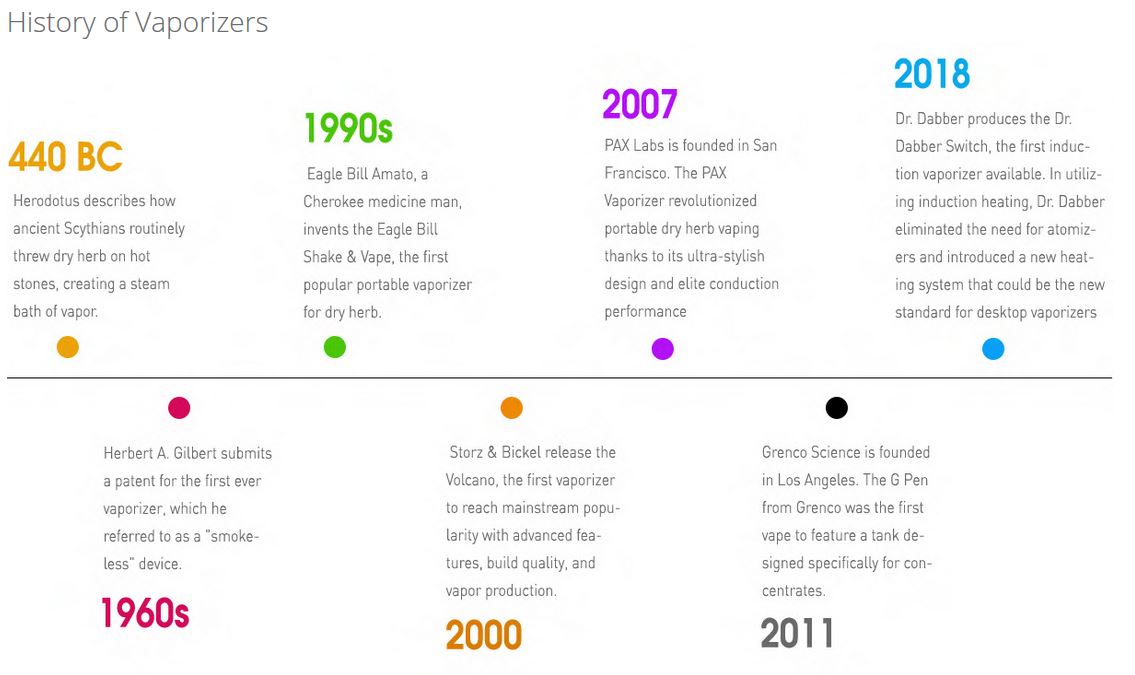
Despite the modern look and feel of today's vaporizers, these devices actually have roots in ancient history. The ancient Greek Historian Herodotus wrote in 440 BC about ancient Scythians throwing dry herbs on hot stones to create steam vapor baths. While it is unknown whether the Scythians were tossing “special” herbs onto the stones, it is still astonishing to know that a device like the DaVinci MIQRO owes its existence, in part, to a practice first documented thousands of years ago.
Vaporizers made their first appearance in 1927 when Joseph Robinson was granted a patent for an “electronic cigarette”. Robinson never managed to commercialize the device, and there might not even have been a prototype, but the desire to introduce vaporizers to the world was now out in the ether. In 1965, Herbert A. Gilbert was granted a patent for the first device that resembles what we think of as e-cigarettes today. Unlike Joseph Robinson, he actually produced prototypes, but just like Joseph Robinson, he wasn’t able to commercialize his work. Gilbert claims the failure of his product was due to companies wanting to wait for the patent to expire rather than license his product, however it is unclear if there was a demand from consumers at the time for electronic cigarettes. For a more in-depth history of e-cigarettes, check out Consumer Advocates For Smoke Free Alternatives' page.
Vaporizer innovation went dormant up until the 1990s when Eagle Bill, a Cherokee medicine man, created what was, at the time, the most commercially successful vaporizer for dry herb, the Eagle Bill Shake and Vape Pipe. Eagle Bill’s goal was to bring vaporization to everyone and his accomplishment paved the way for vaporizers to come. This early vape was incredibly simple. Users placed their dry herb inside of the glass bulb then heated the bottom of the bowl until steam appeared. If users wanted more vapor, they simply gave the piece a gentle shake, hence the name Shake and Vape. The success of the Eagle Bill Shake and Vape Pipe piqued consumers' interest in vaporization, though it would be another decade before the next great wave of innovation washed over the market.

In 1996, Markus Storz developed the Volcano Vaporizer in his basement using a heat gun. Initially developed for the medical community, the Volcano used a “forced air heating system” that propels hot air over material, creating a stream of vapor that is stored in a balloon bag with a mouthpiece attached to it. Two years after Storz completed his first prototype, the Volcano balloon bag patent was registered. The world of smoking would never be the same again.
Storz released the Volcano to European markets in 2000 and consumers raved. Seeing the potential for success, Jugen Bickel joined Storz in 2002, thus forming Storz & Bickel. In 2003, the first 110v devices were built for the American market and the company rapidly expanded. The medical-grade materials and quality management systems in the Volcano led to their company being certified as the first medical-use vaporizers ever, cementing Storz & Bickel as true pioneers in vaporization. To learn more about what Storz & Bickel is all about head over to Leafly for an in-depth history of the Volcano Vaporizer.

While desktop vaporizers boomed, there was a lack of reliable portable options for dry herb users. In 2007, PAX Labs was founded in San Francisco with the goal of creating a sophisticated portable dry herb vaporizer. The first PAX vaporizer opened consumers' eyes to new possibilities for portable dry herb vapes much like the Volcano Vaporizer did for at-home users. With a sleek, discreet design, the PAX opened up portable vaping to a larger audience. Aside from its elegant look and feel (the first model was inspired by the iPod’s simplicity), the PAX provided elite conduction heating to users.
James Monsees, the CEO of PAX Labs, says their inspiration for design was the consumer. “Functionality doesn’t just mean that something turns on or heats up or produces vapor." Said Monsees. "Functionality means that components fit their human interface, that buttons are obvious in their functionality or just plain not there, that knobs and indicators are taken to the absolute level of simplicity and necessity, and all this is done while pushing the bounds of what is possible from an engineering perspective.” To get more of an idea of the initial birth of the PAX vaporizer and what went into the design process you can check out the expanded interview with James Monsees on TechCrunch.

As of 2007, dry herb vaporization was in full swing both portably and for at-home use, but what about the concentrate users? Grenco Science saw that there was a need for more reliable on-the-go concentrate use and decided to do something about it. Founded in 2011, they were the first company to market a vaporizer tank specifically designed for concentrate use. Taking a page from PAX’s design book, all of Grenco’s vaporizers are easily integrated into your daily routine. Simple black silhouettes and no-nonsense operation made it possible for users to explore concentrates. Most, if not all, portable concentrate pens owe their existence to Grenco Science.
The vaporizer industry is an ever-changing landscape, and in 2018 an entirely new heating system burst onto the scene. We’ve briefly highlighted the two major heating systems, conduction and convection, but a third reared its head: induction heating. Induction heating is the process of heating an electrically conducting object through heat generated by eddy currents. This means that heat is generated inside the object itself, instead of an external source, meaning there is no contact at all with a heat source during induction vaping. Dr. Dabber incorporated this heating system into the SWITCH Vaporizer, which uses a large coil to create electromagnetic waves that heat “induction cups” to vaporize either dry herb or concentrate. Induction heating only really exists in the SWITCH, but the success of this desktop vaporizer has opened the possibility of more induction vaporizers being made in the coming years.
Types Of Vapes
The two types of vaporizer are desktop and portable. Desktop vaporizers were the first mass-produced dry herb vaporizers to break into the mainstream. Their efficient heating systems filter out irritants and offer more temperature control over sessions. These units were the first to be used for medicinal purposes.
Portable vaporizers have grown massively in popularity in the last few years. Vape pens are the most discreet and easy-to-use of these units. They heat up quickly and usually offer one-button controls. Portable vaporizers themselves are more powerful than pen-style vapes and offer more control over temperature selection and how the user enjoys the effects and tastes of their preferred materials.
E-Nail vaporizers fall into both categories. These devices offer the huge rips of a concentrate rig, but rather than require the user to torch their materials, they offer convenient electrical heating. Some of these vaporizers are portable with rig-like vapor production.
Desktop Whip Style
Whip Style
- Utilizes a "whip" (rubber tubing) to facilitate vapor consumption
- Inhalation pulls hot air over the dry herb
- First convection-style vaporizes
 Forced Air
Forced Air
- Built-in fan propels hot air into the chamber
- Fills in balloon bag with vapor, making it easy to carry around or share.
- Popularized by the iconic Volcano Vaporizer
 E-Nail
E-Nail
- Eliminates the need for torch
- Allows rig users to zero in on their preferred temperature
- Much safer than using a flame to heat a nail
- Portable models let you take a hit from anywhere
 Multifunctional
Multifunctional
- Offers both whip and balloon filling options
- May include forced air functionality
- All-in-one style unit
 Pen Style
Pen Style
- Rapid heat up time, usually between 5 and 15 seconds
- Pocket-friendly and stealthy
- Some units offer variable voltage
 Portable
Portable
- More powerful than pen style vaporizers
- Higher vapor quality but still portable
- More units offer variable voltage or precision temperature control
Conduction, Convection and Induction Heating
There are three main vaporizer heating methods: Conduction, Convection, and Induction. Your preferred heating method will be based on your vaporizing needs. If you’re an on-the-go user than a conduction vape will give you the fastest heat up time. On the other hand, convection heating releases more flavor and effects with less smoke. Finally, induction offers extremely stable and precise temperature.
Conduction Heating
With conduction vaporization, the herbal material makes direct contact with the heat source to produce vapor. Earlier desktop vaporizers utilized this method, though it is no longer common.

Most wax and pen-style vaporizers utilize this method of vaporization. As the plant material makes direct contact with the heating element, vapor is produced and then inhaled as the user draws through the mouthpiece. Although this method of vaporization results in a quicker heat-up time, it can also lead to combustion if not properly utilized.
Convection Heating
With convection vaporization, the herbal material does not directly touch the heat source. Instead, vaporization is achieved by passing heated air over the material, extracting the active ingredients.

The types of convection vaporizers have expanded greatly since desktop units were the only ones to use this method. There are a multitude of portable vaporizers for both dry herb and concentrate that feature convection heating, giving you unprecedented flavor quality on-the-go. This is the preferred heating method for people wanting to get the fullest flavor from their material.
Induction Heating
Induction heating is similar to convection in that your material is not placed in direct contact with the heating element. Vaporization is achieved by heating an electrically conducting object through heat generated inside the object by eddy currents.
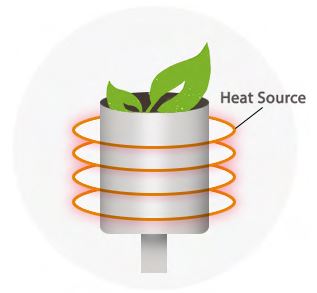
While not as popular as convection or conduction, induction heating is a new vaporization method that gives you the best of both worlds. An incredibly fast heat up time combines with an oxygen-free environment to allow the extraction of 100% of your material’s flavor.
The best induction vaporizer is actually one of the first mass produced units, the Dr. Dabber SWITCH brings a ton of temperature options and can vape both dry herb and concentrates.
Learn more about the Dr. Dabber SWITCH and induction vaping in general from our induction vaporizer specific blog!
What You'll Be Vaping
When it comes to vaporization, there are a variety of materials that can be used, including dry herb, concentrates and e-juices. The material being vaporized will affect which vaporizer you want to use, as not all vapes work with every substance. It is easier to decide on a vape when one knows what they plan to vaporize most frequently.
Dry Herb

- Dried and cured plant material
- Different temperatures release different effects and flavors
- Toggle to 300°F for calmer effects and 430°F for intenser experiences
Concentrates

- Consists of extracted compounds from dry herbs. Enjoy all of the special parts of dry herb without any of the extra, non-usable plant matter
- Much more intense effects than dry herb due to the higher percentage of psychoactive properties
- The ideal temperature range is 325°F for more flavorful experiences. Toggle to 400°F for the full effects of your concentrate
E-Juice/E-Liquid

- Liquid used in electronic cigarettes
- Some flavored liquids may contain nicotine. The vaporizers used for e-juice/-e-liquid are sometimes called “mods” due to their customization capabilities
- Has a higher range of temperatures
Benefits of Using a Grinder
Having a good grind for vaping dry herb is a crucial step to enjoying your vaporizer as it’s meant to be enjoyed. Before vaporizers hit the scene, dry herb users had to use either scissors or their fingers to break up their dry herb, resulting in a ruined household tool or sticky fingers. Thankfully, as vaporizers came about, grinders were already considered a crucial tool for any dry herb session. There are many types of grinders, so we’ll highlight the two most popular. If you’d like more information, check out our grinder page for more info on all the styles of dry herb grinders.
The most common dry herb grinders for vaping are the 2-piece grinder and 4-piece grinder. Two piece grinders are the simplest of those available. They are the most discreet with just a top and bottom piece. Dry herb goes in-between and in a few hand cranks you have finely ground dry herb.
4-piece grinders are a little more in depth. There is a top compartment where dry herb is initially placed. Once ground, dry herb falls into a middle compartment with a screen separating it from the bottom compartment. This screen sifts out pollen from the outside of the dry herb to be saved and stored for later.
The Ideal Vaporizer Grind
If you’re spending a good amount on a tool, you want to make sure you use it correctly, right? To get the most out of your vaporizer, you’ll need to start with a good grind and pack. Grinding your dry herb and loading it the right way allows for vapor to be more abundant, powerful, and smoother than non-ground dry herb. The different heating styles of vaporizers operate better with different consistencies.
Dry herb users who are enjoying a conduction-style vaporizer will want to grind their dry herb as finely as possible. In a conduction-style heating system, the dry herb is heated by direct exposure to the heating element. The finer your grind, the more particles will be exposed to the hot surface. Packing your dry herb tighter inside of the oven will help with the heat transfer and prevent you from having to open your vaporizer and stir dry herbs to make sure they’re all getting vaporized.

For those who are convection-oriented, a medium, fluffy grind is the best way to go. Convection heats your dry herb by hot air, so you’ll want to maximize airflow inside of the chamber. If you use a fine grind, the airflow will be restricted due to the tightness of your dry herb. Finding a grinder that fluffs your dry herb can maximize your convection vape’s capabilities. This leaves hot air with more surface area with which to make contact, thereby ensuring 100% of your dry herb is vaporized. Induction vaporizers don’t have an ideal grind style. The electromagnetic heating field will vaporize any kind of ground dry herb the same way.
Temperature Controls
Controlling the temperature of the vaporizer is key to achieving the most efficient vapor production. There is a range in which the herb will begin to release its active ingredients and if the herb is not warm enough, no vapor forms. Alternatively, if the temperature rises too much, the herb will burn up and defeat the whole purpose of vaporization.
There are vaporizers with digital displays that give you the exact degree that you’re going to be vaping at and those that have pre-set temperatures that feature a different color light assigned to their respective temperature level.
Common Desktop and E Nail Temperature Controls

Desktop vaporizers will have either preset temperatures that were picked by the manufacturer, or precision temperature control, where the temp is chosen by the user.
In the first model of Storz & Bickel’s revolutionary Volcano Vaporizer, a rotary dial let users choose between 9 different settings, allowing for slight changes in temperature. Later on in their lineage, Storz & Bickel produced a digital display model of the Volcano with buttons to allow the user to choose down to the degree what temperature they’d like to vaporize dry herb at, also known as precision temperature control.
The Dr. Dabber SWITCH uses a light meter to help discern temperature levels, with each of the 25 available temperatures having a different number of colored lights assigned to them. Most desktop units have full precision temperature control with a digital readout of your selected temperature.
E-Nails exclusively use precision temperature control to allow users to have full command over their concentrate sessions.
Portable Vaporizer Temperature Controls
Portable vaporizers have a similar distinction between temperature displays as desktop units. Again, this will come down to the personal preference of the user. The two different heating styles are preset temperatures or precision temperature control.
Portable vaporizers with preset temperatures usually feature a one-button control, making them a little easier to use than a precision temperature unit. Users will click the button to power up the vaporizer and toggle through temperature options. Preset temperatures are optimized for the most popular vapor profiles: low temps for flavor, high temps for large clouds. This style is used by those who want quick draws on-the-fly without having to think too much about their temperature choice.
With a precision temperature controlled portable vaporizer you’re able to choose your temperature down to the degree. These vapes will often feature “up” and “down” buttons to help you toggle between temperatures. A wider range of temperature options means you can explore different flavor profiles and customize sessions for flavor, size, or pack density. Most portable vaporizers feature LED displays to help you monitor your temperature.
Vape Pen Temperature Control
Most vape pens will be operated with one-button controls. The small size and sleek designs of these vaporizers are intended to facilitate on-the-go use, and one-button controls provide the quickest and most no-nonsense way of providing vapor.
Just because vape pens have one-button doesn’t mean they can't have multiple temperatures. If a vape pen has one optimized temperature, your experience will be an easy one. Just a few clicks to turn the unit on, then hold down the button as you draw in vapor. For vape pens that have a variable voltage battery that offers multiple temperatures (usually 3) users will turn the vape on, and then click the button a few times to toggle through temperatures.
Vape pens have a limited temperature range compared to portable vaporizers and desktop units because of their focus on portability.
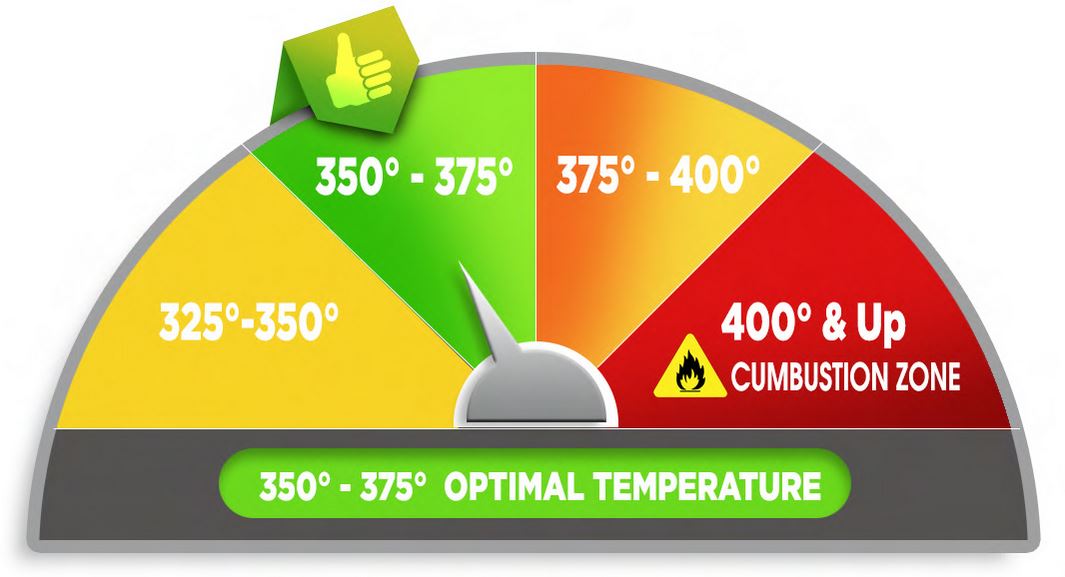
Optimal Temperatures For Vaporizing
The standard temperature for vaporizing dry herb ranges between 350° and 375°, however each vaporizer is different and has their own optimal temperature. Finding the perfect temperature takes a little work and it can vary with the specific material being vaporized. Drier herb will begin vaporizing at a lower temp, whereas a moister herb will require more heat to produce vapor.
Starting at a lower temperature and slowly raising it releases a range of effects as different active ingredients vaporize off with each increase. Everyone has their own personal preference and this should be considered as a guide to find the best temperature to start with.
Heat Up Times
Along with choosing the temperature control, it is good to consider how long the vaporizer will take to go from cold to hot and ready to vape. Each vaporizer's heat up time is different. However, pen-style units are generally the quickest, followed by other portables and then desktop units (which require more power).

Vaporizers that heat up to and maintain a set temperature are more efficient in terms of vapor production. Units that have a push-button and rapid heat up time are more difficult to control, which can easily lead to combustion if you are not careful.
Power Sources
There are several different design factors that determine how a vaporizer's heat source receives power. Ultimately, it will be achieved via either a plug-in or battery.
Plug-In

- Plugs directly into a wall outlet
- Usually only used for desktop vaporizers
- Certain vaporizers, desktop or portable, will feature “pass through” charging, allowing the unit to be used during charging.
Batteries

- Battery-powered vaporizers will usually be Portable or Pen Style units
- Elite portable vaporizers often have easily swappable batteries
- Most vaporizers that are battery powered will have internal batteries housed inside the unit. A few use external batteries, which means you need to press the battery onto the connection to begin heating the vaporizer
Protecting Your Purchase
When it comes to vaporizers, the length of the warranty can vary quite a bit. Generally, desktop vaporizers have the longest warranty, as they are intended to stay in one spot. However, there are portable units such as the Magic Flight that are covered for life. Be certain that the terms of the warranty are well understood prior to making a purchase.
When considering the length of the warranty, also look at what aspect of the vaporizer is actually covered. Not every component (such as glassware or atomizers) is necessarily protected under a warranty. These additional components may be available for purchase if anything ever does break or burn out.
Price Breakdowns
When it comes to vaporizers, the length of the warranty can vary quite a bit. Generally, desktop vaporizers have the longest warranty, as they are intended to stay in one spot. However, there are portable units such as the Magic Flight that are covered for life. Be certain that the terms of the warranty are well understood prior to making a purchase.
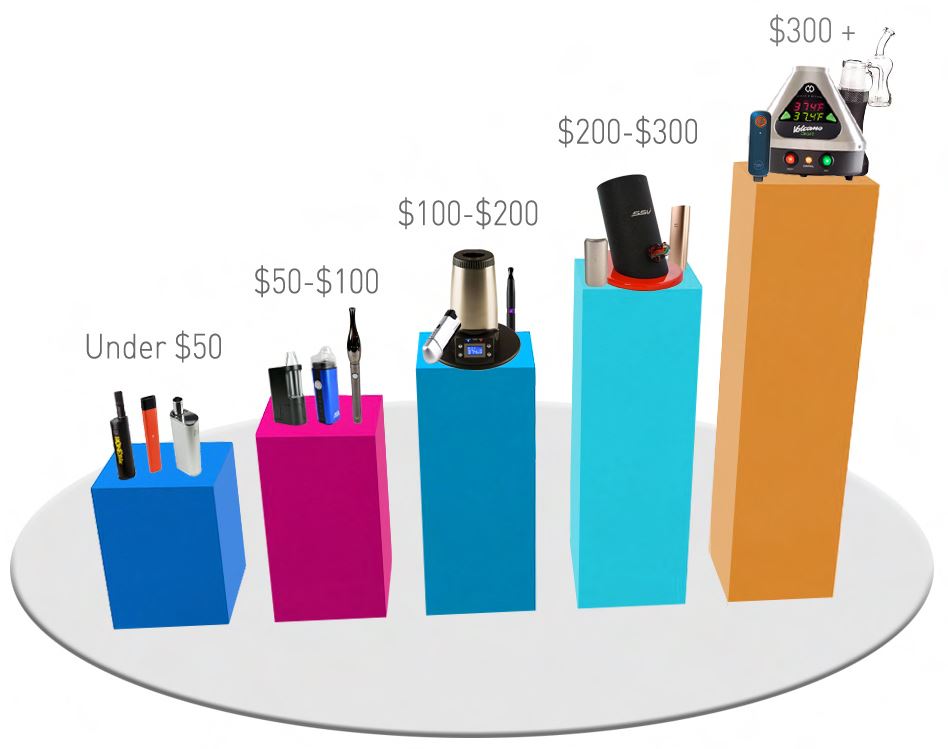
Under $50
If you’re looking for a portable vape or vape pen, this is the category for you. Dry herb or concentrate users can check out our selection of budget-friendly vaporizers that are perfect for on-the-go vaping. The vast majority of these units will be conduction heated.
$50-$100
Users who want more flexibility with temperature and upgraded warranties will find what they need in our $50-$100 category. With the expansion of mobile vaporizer use, top-of-the-line brands have created portable vapes and vape pens that outperform their price range. This is the price range where you’ll see convection heating as an option for your vaporizer.
$100-$200
This is the price range where you will start to see desktop vaporizers available. We have a few in our category, and just because they’re on the cheaper side of desktop units does not mean they can’t perform well. Most vaporizers in this category will utilize convection heating and precision temperature control.
$200-$300
If you’ve enjoyed our lesson on vaporizers and are ready to dive in head first, our $200-$300 range will not let you down. The units available in this category offer you the first “elite” vaporization choices. High-quality build materials, advanced functionality, and beefed up warranties all arrive with your $200-$300 desktop or portable vaporizer.
$300+
Any vaporizer you purchase in the $300+ range will have unparalleled vapor quality, construction, and warranty options. The majority of vaporizers that cost $300+ are desktop units, including the iconic Volcano Vaporizer and the groundbreaking Dr. Dabber SWITCH. It is important to fully understand your vapor needs and how exactly a vaporizer in this price range operates before making a purchase at this level.




